COASY results: Difference between revisions
m (→Table 1) |
m (→Table 1) |
||
| Line 21: | Line 21: | ||
|84 SGSG | |84 SGSG | ||
|- | |- | ||
|- --- | |||
| | |||
|'''Pattern-ID:''' | |'''Pattern-ID:''' | ||
|PKC_PHOSPHO_SITE PS00005 PDOC00005 | |PKC_PHOSPHO_SITE PS00005 PDOC00005 | ||
Revision as of 13:40, 9 June 2007
Structure of Coenzyme A Synthase
Conezyme A Synthase is structurally composed of seven strands, eleven helices and thirteen beta turns (EMBL EBI, 2005) (see Figure 1). Analysis of structurally related proteins (Holm & Sander, 1993) showed a trend for transferase (RCSB, 2007) class proteins with Rossmann class folds (Rossmann, 1973). The Rossmann topologies of fold’s are an alpha-beta class fold that forms a three or more layer beta strand sandwich alternating with alpha helices (beta-alpha-beta-alpha-beta). PFAM classification placed these into either the Cytidylyltransferase or Dephospho-CoA kinase familes, matching the two domains of Coenzyme A Synthase. Those classified under the Dephospho-CoA kinase type were commonly of the P-loop containing nucleotide triphosphate hydrolases (Sanger Institute, 2005) homology whilst those classified as Cytidylyltransferase were of the Tyrosol-Transfer RNA Synthetase (Sanger Institute, 2005). As the majority of the structurally related proteins identified contained the DPCK domain solely, and the motif for a P-loop (Table 1) was identified in the Conzyme A Synthase sequence (Bairoch, Bucher, & Hofmann, 1997) it is suggested that Coenzyme A Synthase is also of the P-loop containing nucleotide triphosphate hydrolases homology of folds.
Table 1
| Pattern-ID: | GLYCOSAMINOGLYCAN PS00002 PDOC00002 | |
| Pattern-DE: | Glycosaminoglycan attachment site | |
| Pattern: | SG.G | |
| Position: | 84 SGSG | |
| Pattern-ID: | PKC_PHOSPHO_SITE PS00005 PDOC00005 | |
| Pattern-DE: | Protein kinase C phosphorylation site | |
| Pattern: | [ST].[RK] | |
| Position: | 3 SDK | |
| Position: | 52 SFR | |
| Position: | 86 SGK | |
| Pattern-ID: | CK2_PHOSPHO_SITE PS00006 PDOC00006 | |
| Pattern-DE: | Casein kinase II phosphorylation site | |
| Pattern: | [ST].{2}[DE] | |
| Position: | 39 SHNE | |
| Position: | 251 TLWE | |
| Position: | 260 SQVE | |
| Pattern-ID: | MYRISTYL PS00008 PDOC00008 | |
| Pattern-DE: | N-myristoylation site | |
| Pattern: | G[^EDRKHPFYW].{2}[STAGCN][^P] | |
| Position: | 82 GISGSG | |
| Position: | 222 GLSEAA | |
| Pattern-ID: | ATP_GTP_A PS00017 PDOC00017 | |
| Pattern-DE: | ATP/GTP-binding site motif A (P-loop) | |
| Pattern: | [AG].{4}GK[ST] | |
| Position: | 82 GISGSGKS | |
| Pattern-ID: | UPF0038 PS01294 PDOC00996 | |
| Pattern-DE: | Uncharacterized protein family UPF0038 signature | |
| Pattern: | G.[LI].R.{2}L.{4}F.{8}[LIV].{5}P.[LIV] | |
| Position: | 136 GTINRKVLGSRVFGNKKQMKILTDIVWPVI |
Figure 1
The secondary structure of Mus musculus with indicated ligand interaction sites (EMBL EBI, 2005).
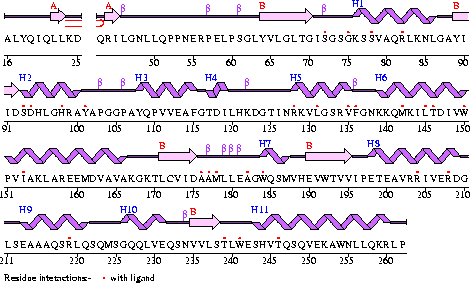
Figure 2
Structural alignment of structurally related proteins to Mus. musculus Coenzyme A Synthase
Localisation Expression of Coenzyme A Synthase
Sequence Conservation of Coenzyme A Synthase
Structural Elements and Functional Binding Sites of Coenzyme A Synthase
Abstract | Introduction | Results | Discussion | Conclusion | Method | References